Jessica James, Ph.D., Rebecca Bryan, Ph.D.; Synopsys; Richie Gill, DPhil, DSc, University of Bath03.19.24
High tibial osteotomy (HTO) is a treatment used for early knee osteoarthritis, but most often for younger patients who may not be as suitable for total knee replacements as older people. One of the key challenges behind designing implants for HTO is the risk of complications and misalignment, which can reduce long-term performance and comfort. One approach to reducing this risk is to use in-silico clinical trials, an emerging method for virtually testing the mechanical safety of personalized implants to better understand safety and performance.
A recent collaboration between the University of Bath, the University of Oxford, the Polytechnic University of Catalonia, Rice University, and the Royal Devon & Exeter NHS Foundation Trust has used in-silico clinical trials as part of development and testing of personalized, 3D-printed HTO plates.1 This tailored approach allows preservation of the knee joint, delivering improved stability and comfort over generic HTO plates, while making the operation to fit them quicker and safer.
The evidence from this study was particularly notable for being the world's first computer-modeled trial to demonstrate the safety of an orthopedic device compared to standard treatment. These in-silico results formed evidence used to gain MHRA approval in the U.K. for ongoing clinical trials, demonstrating the overall value of this method to regulatory workflows and reducing barriers to innovation.
The 3D-printed HTO devices were compared to generic devices to generate evidence to support use of personalized treatments. When combined with existing technologies such as patient-specific 3D-image processing, physics-based simulation, and 3D printing, these types of projects are helping to improve options for novel orthopedic devices for both patients and the medical device industry.
In-silico trials have been used in fields such as drug research, but with some ongoing challenges over consistency and best practice workflows for ensuring the quality of evidence.2 For the medical device industry, it is also necessary for clinical researchers and manufacturers to acquire sufficient clinical data for validation and to ensure modeling technologies and testing frameworks are credible and easily adaptable for different types of devices and interventions.3
However, regulatory pathways are opening up, particularly in the United States, with the FDA developing computational modeling guidance, as well as research into the broader use of modeling and simulation in submissions.4,5 In Europe, there has been a push to also use in-silico trials in the CE marking of medical devices, with clear pathways and harmonized standards to ensure this type of evidence is recognized as credible by assessors.6 While in-silico clinical trials still face these methodological and regulatory hurdles, interventions like the HTO project are showing promising results for how to implement computational simulation into device development and testing.
In-silico clinical trials offer the prospect of improved medical devices reaching the market faster, while dramatically reducing the potential of suffering by patients or animals conventional trials pose. This is a win-win for manufacturers, medical professionals, and, above all, the patient. It is clear from the investment of industry and regulators seen already, the inclusion of in-silico evidence as part of development and regulatory certification will soon be commonplace.
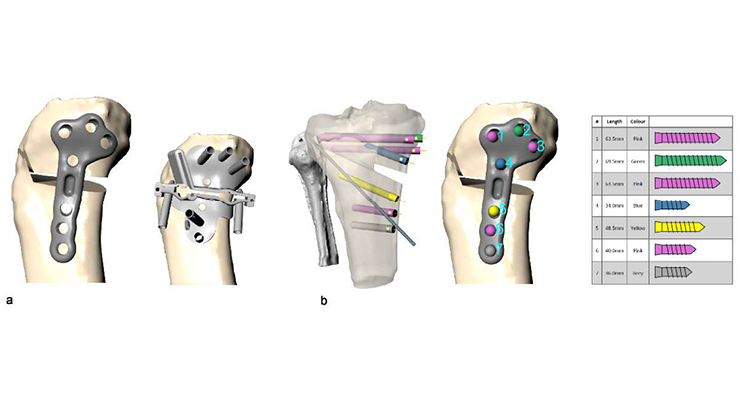
HTO involves creating an opening or closing wedge osteotomy in the proximal tibial to alter the varus alignment; this procedure alters the mechanical axis of the leg, reducing the load in the painful compartment. An osteotomy then involves implanting a stabilizing plate, which can cause pain if improperly fitted. These devices need to have sufficient flexibility to ensure long-term comfort, while also encouraging bone healing. Patient-specific devices offer a potential solution to reduce the risk of poor fitting by creating models and device designs from the actual scan data of the patient. Scanning, building 3D models, simulating mechanical forces, and 3D printing for manufacturing of these devices, therefore, offers a workflow for personalized treatment (Figure 2).
While osteotomy (a.k.a., realignment surgery) is known by knee surgeons to be an effective treatment, many surgeons have concerns about the technical difficulty of achieving the planned correction. A patient-specific digitally planned procedure alleviates these concerns and greatly simplifies the surgical procedure. The study carried out by the University of Bath and its partners compared patient-specific HTO plates and commonly used generic plates through a case-control in-silico clinical trial based off a clinically relevant knee osteoarthritis patient cohort. Thirty knee osteoarthritis patients were CT scanned, with two patients disqualified due to the poor quality of their CT results. The virtual cohort were then created from the segmentation of the CT data, and power analysis carried out using experimentally measured variations in stiffness and strength.
The patient CT data was segmented using Synopsys Simpleware software, and landmarks for regions of interest were identified; required osteotomy correction angles were calculated using MATLAB software. Virtual HTO surgery was simulated using ANSYS SpaceClaim to evaluate changes in the mechanical axis of the knee through an opening wedge osteotomy, with guidance obtained from orthopedic surgeons familiar with these types of procedures.
Following the virtual surgery, each virtual patient was duplicated to test out the osteotomy stabilized using a generic plate, and the other using a personalized plate. The generic plate involved a TomoFix osteotomy plate from DePuy Synthes, with the geometry generated using micro-CT scanning. Simpleware software was used to segment and generate the models. The patient-specific plate was designed using Renishaw plc planning software, with attention paid to the tibia surface and the correction degree for each patient. For the comparison, all simulated knees were tested with both implant types (56 total cases) using ANSYS software.
Computational meshes were then generated to simulate the normal physiological activities a patient would expect to undertake after surgery, such as a fast-walking gait, rising from chairs, and squatting; five key instances were selected for each activity based on the locations of peak tibial contact force. Different simulations and screw configurations were simulated, as well as different lengths of healing stage, focused on the primary outcome of the maximum Von Mises stress within the plates at various bone locations and fatigue levels. This approach necessitated a computationally intense simulation, which involved 500,477 core hours on a high-performance computing cluster.
For the particular study carried out on HTO, the results of the in-silico study were later used to inform a safety trial with five patients, with the goal of demonstrating operations could be quicker and safer while still generating positive clinical outcomes. In the safety trial, one patient was able to take part in a competitive bike ride within six months.7 In addition, a 25-patient-single-arm trial was carried out at the Rizzoli Institute in Bologna, which demonstrated accurate correction for both coronal and sagittal alignment, as well as positive clinical outcomes. A new randomized control trial is now being run in the U.K. to compare patient-specific and traditional methods.8,9
For example, partnerships with on-site 3D-modeling and 3D-printing laboratories at the point of care means designs for personalized devices can be quickly and efficiently created, simulated, manufactured, and used as aids to surgical planning, or as implantable devices. More generally, the process of obtaining regulatory clearance for new devices can be helped by including in-silico tests with virtual populations using specific clinical trial protocols; as well as potentially reducing the burden of clinical testing, this approach can also lower the need for animal testing.
Moreover, there is growing potential for enhancing these workflows through artificial intelligence-enabled methods for speeding up some of the common bottlenecks associated with going from 3D imaging to models, most notably in terms of segmenting regions of interest from anatomies and carrying out routine measurements and other operations on scan data. These types of workflows are already being used to automate designs for surgical cutting guides.10
As with more general uses of AI in healthcare and specific virtual and 3D-printed methods, applying these solutions at the point of care will require regulatory consistency, as well as technical and safety benchmarks. Extensive real-world validation with different populations is also crucial to ensure orthopedic and other medical devices are fit for purpose and safe for patients, with the work being carried out on HTO demonstrating a pathway for combining new technologies with established methods for exploring the suitability of patient-specific procedures and other interventions.
For Kerim Genc of Synopsys, “Leveraging 3D technologies for in-silico trials and additive manufacturing has the potential to revolutionize orthopedic care, offering enhanced treatment personalization and faster, safer regulatory oversight. The pivotal challenge hindering widespread adoption is the automation of workflows. Our strategic focus involves collaborating with industrial and academic partners to develop AI solutions seamlessly integrated into patient and design workflows. Supporting academics, by removing the barriers to software access, is exposing these tools to the emerging generation of engineers who may very well be designing your future implant.”
References
Jessica James, Ph.D., is a business process analyst at the Simpleware Product Group at Synopsys.
Rebecca Bryan, Ph.D., is a product management manager at the Simpleware Product Group at Synopsys.
Richie Gill, DPhil, DSc, is a professor in the Department of Mechanical Engineering at the University of Bath.
A recent collaboration between the University of Bath, the University of Oxford, the Polytechnic University of Catalonia, Rice University, and the Royal Devon & Exeter NHS Foundation Trust has used in-silico clinical trials as part of development and testing of personalized, 3D-printed HTO plates.1 This tailored approach allows preservation of the knee joint, delivering improved stability and comfort over generic HTO plates, while making the operation to fit them quicker and safer.
The evidence from this study was particularly notable for being the world's first computer-modeled trial to demonstrate the safety of an orthopedic device compared to standard treatment. These in-silico results formed evidence used to gain MHRA approval in the U.K. for ongoing clinical trials, demonstrating the overall value of this method to regulatory workflows and reducing barriers to innovation.
The 3D-printed HTO devices were compared to generic devices to generate evidence to support use of personalized treatments. When combined with existing technologies such as patient-specific 3D-image processing, physics-based simulation, and 3D printing, these types of projects are helping to improve options for novel orthopedic devices for both patients and the medical device industry.
In-Silico Clinical Trials
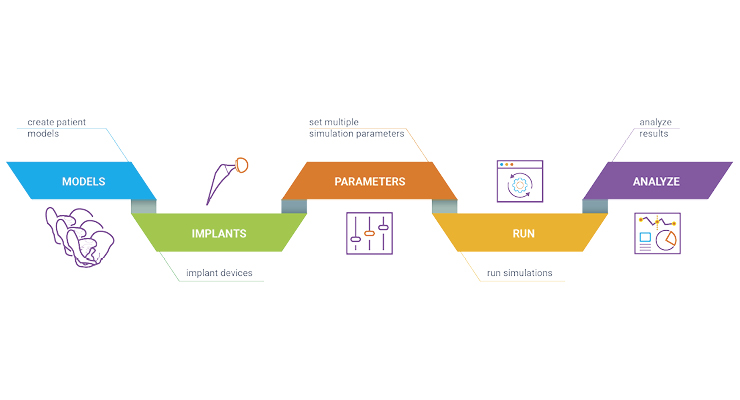
The term “in silico” refers to scientific experiments and testing conducted using computer modeling or computer simulation; in this case, to carry out virtual clinical trials of devices and procedures (Figure 1). With 3D models, it’s possible to simulate multiple surgical outcomes using virtual anatomies of the same individual to test, on a paired basis, mechanical outcomes and failure risk. In this context, each participant is their own trial control. With this method, strict physical clinical trials can be reproduced, adding further evidence for regulatory submissions, and offering potential cost-savings for implant manufacturers.In-silico trials have been used in fields such as drug research, but with some ongoing challenges over consistency and best practice workflows for ensuring the quality of evidence.2 For the medical device industry, it is also necessary for clinical researchers and manufacturers to acquire sufficient clinical data for validation and to ensure modeling technologies and testing frameworks are credible and easily adaptable for different types of devices and interventions.3
However, regulatory pathways are opening up, particularly in the United States, with the FDA developing computational modeling guidance, as well as research into the broader use of modeling and simulation in submissions.4,5 In Europe, there has been a push to also use in-silico trials in the CE marking of medical devices, with clear pathways and harmonized standards to ensure this type of evidence is recognized as credible by assessors.6 While in-silico clinical trials still face these methodological and regulatory hurdles, interventions like the HTO project are showing promising results for how to implement computational simulation into device development and testing.
In-silico clinical trials offer the prospect of improved medical devices reaching the market faster, while dramatically reducing the potential of suffering by patients or animals conventional trials pose. This is a win-win for manufacturers, medical professionals, and, above all, the patient. It is clear from the investment of industry and regulators seen already, the inclusion of in-silico evidence as part of development and regulatory certification will soon be commonplace.
Procedure
Knee osteoarthritis is becoming more common, and while knee replacement demand is growing, this intervention is only suitable for end-stage disease, and due to its non-reversible nature, is less well-suited to younger patients. Complications to these surgeries include impact on function, the need for revision, and general patient dissatisfaction. In this context, alternative osteoarthritis treatments are crucial to expand, with HTO providing a reliable option for knee preserving treatment for early-stage and even more advanced osteoarthritis.HTO involves creating an opening or closing wedge osteotomy in the proximal tibial to alter the varus alignment; this procedure alters the mechanical axis of the leg, reducing the load in the painful compartment. An osteotomy then involves implanting a stabilizing plate, which can cause pain if improperly fitted. These devices need to have sufficient flexibility to ensure long-term comfort, while also encouraging bone healing. Patient-specific devices offer a potential solution to reduce the risk of poor fitting by creating models and device designs from the actual scan data of the patient. Scanning, building 3D models, simulating mechanical forces, and 3D printing for manufacturing of these devices, therefore, offers a workflow for personalized treatment (Figure 2).
While osteotomy (a.k.a., realignment surgery) is known by knee surgeons to be an effective treatment, many surgeons have concerns about the technical difficulty of achieving the planned correction. A patient-specific digitally planned procedure alleviates these concerns and greatly simplifies the surgical procedure. The study carried out by the University of Bath and its partners compared patient-specific HTO plates and commonly used generic plates through a case-control in-silico clinical trial based off a clinically relevant knee osteoarthritis patient cohort. Thirty knee osteoarthritis patients were CT scanned, with two patients disqualified due to the poor quality of their CT results. The virtual cohort were then created from the segmentation of the CT data, and power analysis carried out using experimentally measured variations in stiffness and strength.
The patient CT data was segmented using Synopsys Simpleware software, and landmarks for regions of interest were identified; required osteotomy correction angles were calculated using MATLAB software. Virtual HTO surgery was simulated using ANSYS SpaceClaim to evaluate changes in the mechanical axis of the knee through an opening wedge osteotomy, with guidance obtained from orthopedic surgeons familiar with these types of procedures.
Following the virtual surgery, each virtual patient was duplicated to test out the osteotomy stabilized using a generic plate, and the other using a personalized plate. The generic plate involved a TomoFix osteotomy plate from DePuy Synthes, with the geometry generated using micro-CT scanning. Simpleware software was used to segment and generate the models. The patient-specific plate was designed using Renishaw plc planning software, with attention paid to the tibia surface and the correction degree for each patient. For the comparison, all simulated knees were tested with both implant types (56 total cases) using ANSYS software.
Simulation
Finite element simulation enables realistic testing of mechanical properties, such as stress and strain (Figure 3). The models for this study were designed to represent the screw and plate, as well as contact interactions between different parts of the model. Patient-specific material properties were applied to the simulation from each patient’s CT data, while other factors were determined by experimental testing and default software sections. The progression of bone healing was modeled by increasing the Young’s modulus of the osteotomy region, based on literature values.Computational meshes were then generated to simulate the normal physiological activities a patient would expect to undertake after surgery, such as a fast-walking gait, rising from chairs, and squatting; five key instances were selected for each activity based on the locations of peak tibial contact force. Different simulations and screw configurations were simulated, as well as different lengths of healing stage, focused on the primary outcome of the maximum Von Mises stress within the plates at various bone locations and fatigue levels. This approach necessitated a computationally intense simulation, which involved 500,477 core hours on a high-performance computing cluster.
Results
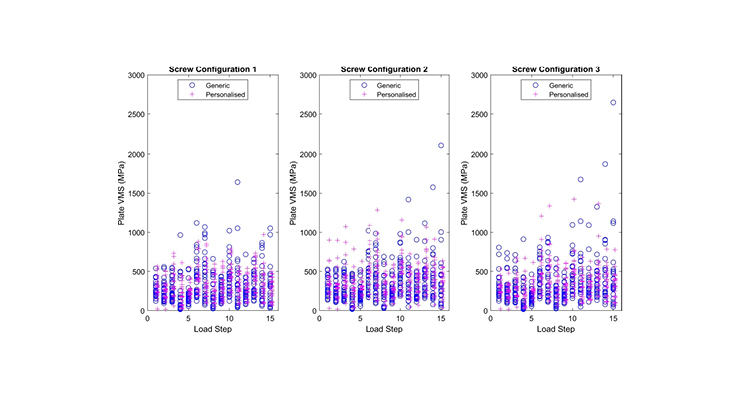
Testing results demonstrated the effect of the screw configuration on the Von Mises stress in each type of plate was not statistically significant, but the effect of the healing stage was very dramatic in terms of reducing the maximum stresses in both generic and patient-specific plates. In this context, more significant differences in inter-fragmentary movement between the devices were observed 12 weeks postoperatively due to increased micro-motion. Physiological loading results showed the personalized HTO plates had slightly larger stress values than the generic plates, but without enough of a difference to be a concern; the personalized plates had no difference in their failure risk compared to the generic model and were more mechanically efficient and less stiff. These results provide promising evidence that 3D-printed plates from patient-specific image data can help expand the use of HTO surgery.Impact and Implementation
The use of in-silico workflows generally helps support the development of personalized orthopedic devices while reducing potential risks. In cases involving clinical trials for personalized plates, virtual testing has the potential to cut down on time and costs for device manufacturers. This type of testing and use of simulation also offers opportunities for efficiencies when designing and manufacturing personalized devices, as different iterations and surgical scenarios can be digitally planned without the use of higher-cost materials. It is worth noting, however, in-silico testing does not replace traditional methods such as clinical patient trials or benchtop tests but provides a valuable additional resource for companies to better understand products and reduce risk through increased data backed by patient imaging.For the particular study carried out on HTO, the results of the in-silico study were later used to inform a safety trial with five patients, with the goal of demonstrating operations could be quicker and safer while still generating positive clinical outcomes. In the safety trial, one patient was able to take part in a competitive bike ride within six months.7 In addition, a 25-patient-single-arm trial was carried out at the Rizzoli Institute in Bologna, which demonstrated accurate correction for both coronal and sagittal alignment, as well as positive clinical outcomes. A new randomized control trial is now being run in the U.K. to compare patient-specific and traditional methods.8,9
Conclusions
The breakthrough use of in-silico trials for orthopedic safety testing and informing clinical trials is also highly significant for demonstrating the value of this method to device manufacturers. In-silico trials offer orthopedic companies the potential for more wide-ranging evaluation and better understanding of the impact of the design on performance, without risking patient suffering, as well as significant time and cost savings over traditional regulatory evidence generation routes. This innovative application makes the mass manufacture and implantation of customized orthopedic devices a reality, with benefits to all involved.For example, partnerships with on-site 3D-modeling and 3D-printing laboratories at the point of care means designs for personalized devices can be quickly and efficiently created, simulated, manufactured, and used as aids to surgical planning, or as implantable devices. More generally, the process of obtaining regulatory clearance for new devices can be helped by including in-silico tests with virtual populations using specific clinical trial protocols; as well as potentially reducing the burden of clinical testing, this approach can also lower the need for animal testing.
Moreover, there is growing potential for enhancing these workflows through artificial intelligence-enabled methods for speeding up some of the common bottlenecks associated with going from 3D imaging to models, most notably in terms of segmenting regions of interest from anatomies and carrying out routine measurements and other operations on scan data. These types of workflows are already being used to automate designs for surgical cutting guides.10
As with more general uses of AI in healthcare and specific virtual and 3D-printed methods, applying these solutions at the point of care will require regulatory consistency, as well as technical and safety benchmarks. Extensive real-world validation with different populations is also crucial to ensure orthopedic and other medical devices are fit for purpose and safe for patients, with the work being carried out on HTO demonstrating a pathway for combining new technologies with established methods for exploring the suitability of patient-specific procedures and other interventions.
For Kerim Genc of Synopsys, “Leveraging 3D technologies for in-silico trials and additive manufacturing has the potential to revolutionize orthopedic care, offering enhanced treatment personalization and faster, safer regulatory oversight. The pivotal challenge hindering widespread adoption is the automation of workflows. Our strategic focus involves collaborating with industrial and academic partners to develop AI solutions seamlessly integrated into patient and design workflows. Supporting academics, by removing the barriers to software access, is exposing these tools to the emerging generation of engineers who may very well be designing your future implant.”
References
- tinyurl.com/27j7vpr5
- tinyurl.com/4yy3au4s
- tinyurl.com/2mfvarek
- tinyurl.com/2vkuumma
- tinyurl.com/89kmsswk
- tinyurl.com/3pntsur5
- tinyurl.com/yva6fw9s
- tinyurl.com/5n6mccvh
- tinyurl.com/4cye5c65
- tinyurl.com/2n5pyfuc
Jessica James, Ph.D., is a business process analyst at the Simpleware Product Group at Synopsys.
Rebecca Bryan, Ph.D., is a product management manager at the Simpleware Product Group at Synopsys.
Richie Gill, DPhil, DSc, is a professor in the Department of Mechanical Engineering at the University of Bath.