David C. Kelman, President, Kelman & Company Consulting LLC 05.29.18
The healthcare market has been changing over the past 30 years and the speed of change is increasing significantly. During this timeframe, great advances have been made to patient care through new surgical interventions and medical devices. Early on in my career, the focus was to enhance medical technology so patient outcomes would improve. This was the primary driver and there was limited concern about the manufacturing cost of a device. Over time, this focus has changed.
For the past 40 years, the rate of healthcare spending has grown at twice the rate of per capita income.1 This is not only a fact in the United States, but also for most of the developed world. We have all seen it and, more importantly, felt it in our wallets. The cost of our medical insurance premiums has steadily increased alongside our deductible limits. These increased costs have placed increased pressure on all aspects of the healthcare network. Reported in a 2016 Forbes article,1 the Affordable Care Act of 2010 “restricts the growth of real per capita Medicare spending to no more than the rate of growth of real GDP per capita plus about one half of a percent—forever.” Provisions were made to make it very difficult to increase the spending for Medicare. The net effective will be reduced payments to the providers and then continued, if not increased, pressure on the medical device manufacturers.
The job of the product development engineer has already been difficult and will become even more arduous. To succeed, the product development engineer is going to have to be so much more than in the past. They will have to act as an independent businessperson running their own firm. In other words, they will need to understand all aspects of running a business, as every decision they make impacts the financial performance of their organization. While in the past, high gross margins enabled a poor-performing offering to be hidden by other products, the same cannot be said in today’s cost conscious environment. Further, today’s successful orthopedic products have the potential to have a 20- to 30-year commercial lifecycle, in stark contrast to the five to six years of the 1980s. The design decisions the development engineer makes in the concept and early development stages are very much locked into the life of the product. This includes the marketability, sales potential, manufacturing, inventory, and distribution costs for a product with that 20-plus years potential commercial lifecycle.
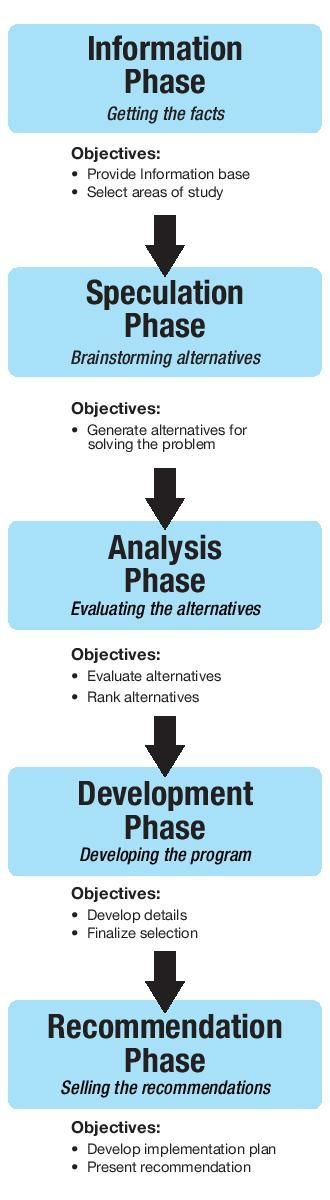
Figure 1: Phases in the Value Engineering job plan.2
For years, companies have always worked on cost reductions of products after the design has been finalized. These are currently and will remain ongoing efforts, but the ability to make significant impacts on reducing costs is limited by the design constraints. Unit cost, inventory cost, and instrumentation costs are often the only financial considerations utilized in the stage gate development review process, but this does not provide a complete picture of the financial profit loss statement of the product once it has been commercialized. Selling, general, and administrative (SG&A) costs for some medical device companies represents one and a half times the cost of manufacture. A major contributor to the SG&A costs are the various field inventory locations, instrumentation (that remains on the books of the manufacturer), and the shipping costs for the instruments and/or implants to and from the hospital.
To develop a technically successful product, as well as a financially successfully one, the product development engineer will need to consider a full pro forma statement. As the development engineer creates various design iterations and assesses the viability of those designs, they must do the same with the full financial view of the new product to optimize both the technical and financial aspects of it. The finance department will have to become an active partner early and throughout the iterative development process with a complete financial model that itself will have to be iterative. But how is this accomplished?
A technique developed in the post-World War II timeframe at GE by Lawrence Miles—Value Engineering—is one method that can be utilized. The definition of Value Engineering is “To deliver the required functions of a component or product at lowest cost while meeting quality, performance, and reliability specifications.”2 I had the opportunity to work with Tom Snodgrass, a lifelong colleague of Miles, while I was a student at the University of Wisconsin and was able to learn some of the basic concepts of Value Engineering through numerous conversions. These concepts have stayed with me throughout my career. The process of Value Engineering should be a very familiar process to the development engineer, but with a focus on cost and function versus function exclusively (Figure 1).
Information Phase
This step represents what the development engineer has traditionally performed to understand the functional requirements. The engineer must take a moment to ask, “What function must the product perform?” without jumping to a solution. Using a total knee system as an example, what is the range of femurs the system addresses? Obtain the necessary criteria that will be utilized to assess the possible solutions, technical requirements, marketing desires, and cost to service.
Speculation Phase
Develop several different scenarios regarding the rationale and reason for various numbers of sizes. Is each change in size uniform; for example, is it a fixed 4-mm change/size? Or is the change in size progressive and relative to the increasing size of the implant? What other possible change in size alternatives can you brainstorm? Each method could potentially lead to a different number of sizes to address the orthopedic application.
Analysis Phase
Evaluate the various solutions developed, utilizing the criteria gathered during the Information Phase, weighing each of the criteria appropriately. During this phase, several of the alternatives would be selected for further refinement and development.
Development Phase
The selected alternative solutions from the Analysis Phase would be further developed to ensure a more robust appraisal can be performed. For example, total knee system A has 10 different sizes, while total knee system B has seven. This difference impacts the number of tibial trays, tibial inserts, and instruments required for each system. The cost should not be viewed as only the manufacturing expense for each femoral component or the total inventory cost, but more of the total cost to service the account. In other words, consider what would be used to evaluate the performance of the system post-launch.
Introductions of new systems are often limited by available capital and decisions to limit inventory levels in the field. Alternative solutions with lower capital costs for instrumentation and inventory requirements offer the possibility of being able to provide more sets to the field at the same cost. One solution has been the introduction of “loaners”—the system is moved into and out of the hospital for each case. Shipping and handling costs, however, are not insignificant. The larger the system, the greater the cost. This distribution model has contributed greatly to the high SG&A costs for many medical device manufacturers. It is at this phase where the development engineer can have a significant impact either positively or negatively.
Recommendation Phase
Obtaining approval from all the key stakeholders—surgeon designers, marketing, and corporate management—is critical. It is important the criteria and the objectives are determined prior to developing the final solutions. Getting alignment and acceptance from the key stakeholders at the beginning provides the foundation for the project. Therefore, when the recommended solution is provided, it is less likely to become derailed by the interests of one stakeholder.
Conclusion
As the pressures from rising healthcare costs continue, the requirements for the development engineer to innovate, develop new products, and optimize the commercial aspects of those products will continue to become more daunting. The development engineer will have to examine their projects with a broader lens as to the requirements of what a successful product is and their influence upon it. Greater support of the finance department will be required to simulate the various configurations to gain greater insight on the impact of the design considerations and alternatives on the short- and long-term commercial viability of the product over its anticipated 20-plus year lifecycle. Value Engineering is just one possible tool the product development engineer can use.
References
http://bit.ly/odt180610
http://bit.ly/odt180611
David C. Kelman, president of Kelman & Company Consulting LLC, is a licensed professional engineer who has managed and led design and development teams on three different continents for major orthopedic companies. He is a creative innovator whose broad scope of solutions has resulted in two unique start-up businesses, commercialization of 40 product platforms, and 50 U.S. patents awarded. As a multifaceted problem solver, he incorporates technological and business sophistication across diverse environments, including product design/software/analytics/supply chain/technical marketing and sales. This unique mix of technical, engineering, and business expertise has resulted in successful business strategies, and the introduction of cutting-edge technologies with financial viability.
For the past 40 years, the rate of healthcare spending has grown at twice the rate of per capita income.1 This is not only a fact in the United States, but also for most of the developed world. We have all seen it and, more importantly, felt it in our wallets. The cost of our medical insurance premiums has steadily increased alongside our deductible limits. These increased costs have placed increased pressure on all aspects of the healthcare network. Reported in a 2016 Forbes article,1 the Affordable Care Act of 2010 “restricts the growth of real per capita Medicare spending to no more than the rate of growth of real GDP per capita plus about one half of a percent—forever.” Provisions were made to make it very difficult to increase the spending for Medicare. The net effective will be reduced payments to the providers and then continued, if not increased, pressure on the medical device manufacturers.
The job of the product development engineer has already been difficult and will become even more arduous. To succeed, the product development engineer is going to have to be so much more than in the past. They will have to act as an independent businessperson running their own firm. In other words, they will need to understand all aspects of running a business, as every decision they make impacts the financial performance of their organization. While in the past, high gross margins enabled a poor-performing offering to be hidden by other products, the same cannot be said in today’s cost conscious environment. Further, today’s successful orthopedic products have the potential to have a 20- to 30-year commercial lifecycle, in stark contrast to the five to six years of the 1980s. The design decisions the development engineer makes in the concept and early development stages are very much locked into the life of the product. This includes the marketability, sales potential, manufacturing, inventory, and distribution costs for a product with that 20-plus years potential commercial lifecycle.

Figure 1: Phases in the Value Engineering job plan.2
To develop a technically successful product, as well as a financially successfully one, the product development engineer will need to consider a full pro forma statement. As the development engineer creates various design iterations and assesses the viability of those designs, they must do the same with the full financial view of the new product to optimize both the technical and financial aspects of it. The finance department will have to become an active partner early and throughout the iterative development process with a complete financial model that itself will have to be iterative. But how is this accomplished?
A technique developed in the post-World War II timeframe at GE by Lawrence Miles—Value Engineering—is one method that can be utilized. The definition of Value Engineering is “To deliver the required functions of a component or product at lowest cost while meeting quality, performance, and reliability specifications.”2 I had the opportunity to work with Tom Snodgrass, a lifelong colleague of Miles, while I was a student at the University of Wisconsin and was able to learn some of the basic concepts of Value Engineering through numerous conversions. These concepts have stayed with me throughout my career. The process of Value Engineering should be a very familiar process to the development engineer, but with a focus on cost and function versus function exclusively (Figure 1).
Information Phase
This step represents what the development engineer has traditionally performed to understand the functional requirements. The engineer must take a moment to ask, “What function must the product perform?” without jumping to a solution. Using a total knee system as an example, what is the range of femurs the system addresses? Obtain the necessary criteria that will be utilized to assess the possible solutions, technical requirements, marketing desires, and cost to service.
Speculation Phase
Develop several different scenarios regarding the rationale and reason for various numbers of sizes. Is each change in size uniform; for example, is it a fixed 4-mm change/size? Or is the change in size progressive and relative to the increasing size of the implant? What other possible change in size alternatives can you brainstorm? Each method could potentially lead to a different number of sizes to address the orthopedic application.
Analysis Phase
Evaluate the various solutions developed, utilizing the criteria gathered during the Information Phase, weighing each of the criteria appropriately. During this phase, several of the alternatives would be selected for further refinement and development.
Development Phase
The selected alternative solutions from the Analysis Phase would be further developed to ensure a more robust appraisal can be performed. For example, total knee system A has 10 different sizes, while total knee system B has seven. This difference impacts the number of tibial trays, tibial inserts, and instruments required for each system. The cost should not be viewed as only the manufacturing expense for each femoral component or the total inventory cost, but more of the total cost to service the account. In other words, consider what would be used to evaluate the performance of the system post-launch.
Introductions of new systems are often limited by available capital and decisions to limit inventory levels in the field. Alternative solutions with lower capital costs for instrumentation and inventory requirements offer the possibility of being able to provide more sets to the field at the same cost. One solution has been the introduction of “loaners”—the system is moved into and out of the hospital for each case. Shipping and handling costs, however, are not insignificant. The larger the system, the greater the cost. This distribution model has contributed greatly to the high SG&A costs for many medical device manufacturers. It is at this phase where the development engineer can have a significant impact either positively or negatively.
Recommendation Phase
Obtaining approval from all the key stakeholders—surgeon designers, marketing, and corporate management—is critical. It is important the criteria and the objectives are determined prior to developing the final solutions. Getting alignment and acceptance from the key stakeholders at the beginning provides the foundation for the project. Therefore, when the recommended solution is provided, it is less likely to become derailed by the interests of one stakeholder.
Conclusion
As the pressures from rising healthcare costs continue, the requirements for the development engineer to innovate, develop new products, and optimize the commercial aspects of those products will continue to become more daunting. The development engineer will have to examine their projects with a broader lens as to the requirements of what a successful product is and their influence upon it. Greater support of the finance department will be required to simulate the various configurations to gain greater insight on the impact of the design considerations and alternatives on the short- and long-term commercial viability of the product over its anticipated 20-plus year lifecycle. Value Engineering is just one possible tool the product development engineer can use.
References
http://bit.ly/odt180610
http://bit.ly/odt180611
David C. Kelman, president of Kelman & Company Consulting LLC, is a licensed professional engineer who has managed and led design and development teams on three different continents for major orthopedic companies. He is a creative innovator whose broad scope of solutions has resulted in two unique start-up businesses, commercialization of 40 product platforms, and 50 U.S. patents awarded. As a multifaceted problem solver, he incorporates technological and business sophistication across diverse environments, including product design/software/analytics/supply chain/technical marketing and sales. This unique mix of technical, engineering, and business expertise has resulted in successful business strategies, and the introduction of cutting-edge technologies with financial viability.