Michael Barbella , Managing Editor02.17.16
The letter was short, blunt and non-apologetic. In it, Raymond L. Horwood, M.D.—a 2011 physician of the year—severs ties with a Cleveland Clinic hospital over a mandate limiting orthopedic implant options.
Horwood’s umbrage is clearly evident as he informs patients of his decision, blaming the move on a Cleveland Clinic directive to use joint replacement parts made only by Stryker Corp. or Zimmer Holdings Inc. In his letter late last summer, Horwood—an orthopedic surgeon—avoids critiquing the two companies’ implant quality, but hints that switching allegiances from DePuy products could negatively impact treatment.
“The Cleveland Clinic Foundation has instituted cost-cutting measures which limit the implant options for joint replacement surgery to just two vendors...Stryker and Zimmer,” Horwood’s letter read. “Dr. Horwood has 28 years of experience with DePuy J&J implants which, in his hands, have resulted in excellent clinical outcomes. Dr. Horwood does not feel in good conscience that he can jeopardize his patients by changing to an unfamiliar implant system simply to save money for the hospital system.”
Indeed, money played a key role in Horwood's breakup with the Cleveland Clinic. Physicians and hospital administrators have developed an adversarial relationship over the last half-decade as they’ve grappled with ways to provide quality healthcare at a lower cost. The challenge has frustrated both sides, though doctors have become particularly resentful of managers’ bottom-line tunnel vision.
Finances, however, may only be partly responsible for Horwood’s decision. Other likely contributors were product familiarity and, ultimately, resistance to change.
Physicians, like most human beings, are creatures of habit, preferring the familiar over the unknown. Most clinicians, for example, resisted the idea of electronic medical records in the late 1990s, deeming the time-saving tool an impersonal addition to their practices and an affront to the profession.
Many doctors have developed a similar mindset regarding medical products and equipment. Horwood, as he admitted in his letter, is a DePuy devotee; others are disciples of certain procedures (robotic surgery, for instance) or materials (trabecular metal, polyethylene, stainless steel, ceramics, etc.), and some have favorite tools.
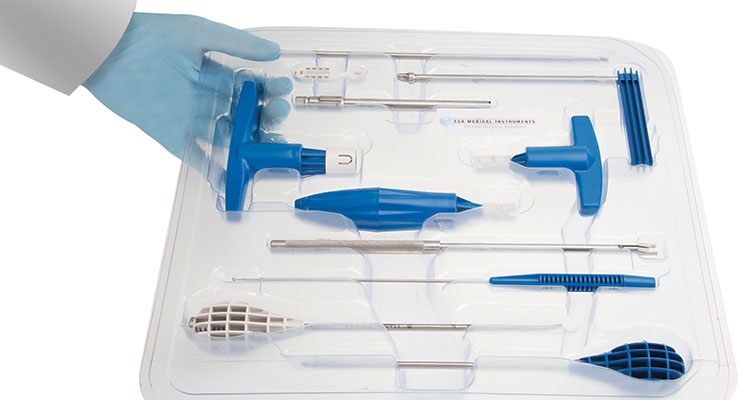
Orthopedic surgeons have traditionally endorsed reusable instruments and surgical trays, but competition, healthcare reform, accuracy concerns, and—perhaps most importantly—cost, are engendering change in the operating room. Newly minted single-use, torque-limiting instruments and kits are becoming more prevalent as hospitals seek procedural efficiency, streamlined inventory, environmental harmony, and reduced cost.
While single-use instruments and kits have been a staple of many medical procedures for decades (cardiac rhythm management and neuromodulation, among others) they are a relatively new development in the orthopedic industry, particularly in the spinal sector.
Companies like Safe Orthopaedics, ECA Medical Instruments Inc. and Flower Orthopedics have developed single-use instruments for spinal surgery that promise to “eliminate unnecessary delays” as well as the need for expensive sterilization cycles. ECA’s disposable ratchet (the Model 300 Secure-Fast system) is designed to reduce surgical costs by more than $1,000 per procedure—a factor that will surely appeal to clinicians and payers striving to balance quality care and price value.
The global market for disposable medical supplies (instruments included) is forecast to grow 4.1 percent annually over the next two years, fueled by healthcare reform, an aging population, and a sharp focus on infection prevention. But convenience, cost, inventory management, and improved productivity will drive growth too, as more hospitals and surgeons realize their economic and clinical value.
To better understand the market forces responsible for single-use instrumentation as well as design trends and promising technological developments in surgical tools, Orthopedic Design & Technology spoke to a handful of manufacturing professionals and suppliers over the last few weeks. They included:
Michael Barbella: Please discuss the trends in orthopedic instrumentation and delivery systems.
Toby Buck: Trends that all contract manufacturing organizations (CMOs)—those like Greatbatch, Tecomet, Orchid—would say that the industry right now is under extreme stress because of downward cost pressures and that is directly affecting not only instruments but delivery systems, bundled services and implants. And that’s playing out obviously at our clients but it’s certainly trickled down and conditions the behavior of all of our income statements presently. So I think cost is the order of the day today, I think procurement organizations are the ones that are calling the shots internal to the OEMs out of necessity. I think all of us have to adjust and are adjusting to this context in a new operating paradigm. We have to not only continue to stay with the OEMs and serve them how they need to be served, but we have to get ahead of them to amplify our value proposition.
Mark Kemp: Tecomet is one of the largest contract manufacturers in the world and services several different market segments within the orthopedic industry. We continue to see the need for different levels of procedural innovation across all of these segments. To further address these opportunities, we offer multiple processing technologies that can result in value-added cost savings to our global customer base. Recently, our customers have increasingly requested the need for further instrument set cost reductions. Our Total Solution business approach attempts to manage these requests by working with the customer to reconfigure these sets to contain the essential core instruments. Additional innovation and cost reductions are being achieved via supply chain optimization opportunities and vendor-managed inventory is being further utilized.
Jack Neenan: MI (minimally invasive) surgical procedures are driving orthopedic instrument requirements. The benefit to MI procedures is less invasive and quicker recovery times for patients. On the case and tray side we are seeing some of our OEM customers looking to move away from using blue plastic wrap on vented cases and taking those same vented aluminum or stainless steel cases and putting them inside a “hard” pressure vented case. The “hard vented” case does away with the need for blue plastic wrap.
James Schultz: Each OEM implant firm wants to win more share of the fast-growth ambulatory surgery center (ASC) or outpatient market. It is very big and growing at 15 percent or more per year. Over 5,700 ASCs exist in the United States alone and 18 percent of their procedures are for orthopedics/spine. Who wants to go to a hospital? A day trip to the ASC is preferred and less expensive. More and more procedures are being approved yearly by the FDA [U.S. Food and Drug Administration] and payers are reimbursing these procedures at an increasing clip. OEMs will win if they have an implant solution that provides excellent outcomes and measured economic value.
The biggest issue facing every instrument manufacturer, implant OEM and end-user customer is cost. Each one is addressing how to drive out both upfront and life cycle costs to serve both the ASC and hospital market segments. Older business models are not sustainable. Moving forward there will be a broad application of lower-cost instruments and procedural kits, and especially the adoption of single-procedure/disposable instrumentation. Disposable instruments and procedure kits solve many active pain points today: They lower upfront and life cycle cost, reduce the risk of infection, are pristine and ready for surgery, curb inventory hassles, and are in perfect calibration to secure every orthopedic/spine implant.
Barbella: How has orthopedic instrumentation evolved over the last five to 10 years, and what major changes have you witnessed?
Buck: The orthopedic industry has learned from past sins of poor inventory management in the field and how that is supported not only logistically but also in the definition of launch quantities. I think the launches we experienced in the past as an industry with large hip and knee systems or large spine systems, for example, with instrumentation sets being deployed in the field, are not going to happen in the same manner they once did. The change is ever-present in terms of the cadence on launches for programs in the dominant segments we serve in orthopedics—hips, knees, spine, sports medicine, trauma and extremities. Over the last five to 10 years, there have been some major launches by all major OEMs but you have a parallel market implication associated with consolidation both at the OEM level and at the Tier 1 CMO level that has forced portfolio rationalization at the OEM level and, correspondingly, forced CMO consolidation to amplify the value proposition on the sourcing side of the fence for instrumentation, delivery systems and implants.
Another thing we need to do as an industry to get cost out of the game is…if you’re truly going to be a strategic partner, you have to agree between OEM and CMO, particularly with instrument applications, to embrace true design for manufacturing (DFM), true upstream feature-based pricing, and address the issues associated or attached to tolerancing that isn’t necessary in the actual utility application of the instrument or instrument set. You can get a tremendous amount of cost out through a pure application of DFM doctrine, feature-based pricing, and common sense applied to topographical tolerancing in instrumentation and delivery systems.
When you think about the configuration, the various topography, or the various dimensional characteristics of an instrument, there are radiuses and different features regarding the manner in which an instrument has tolerances that are associated with the finished machined product that are just too tight. The tighter a tolerance is—and this is common sense—it amplifies cost. There’s a parallel sensitivity that we all need to be aware of or mindful of as well—instrumentation today is treated in terms of regulatory compliance in the same manner that implants are. How can we expect as an industry to continue to impose a severe downward cost pressure on instrumentation and how can that coexist with amplified regulatory compliance, while CMS [the Center for Medicare and Medicaid Services] is compressing reimbursement thresholds? I don’t know how those things co-exist. And that’s causing the extreme downward cost pressure and stress on the industry that we’re experiencing right now. A related issue that has been coming for some time is less than effective instrument set inventory management in the field leading to too many sets in the field. Instrumentation is something that is not necessarily sold. Instrumentation is provided to the distribution infrastructure for the OEMs. Their distributors may or may not acquire some of it, but generally speaking instrumentation is treated as though it is tooling in the field for the OEM. That’s another part of the economic solution here that we all have to be sensitive to and ever-mindful of to help continue to find a way to amplify our value proposition to the marketplace as a CMO for integrated instrument set and bundled service sourcing.
Kemp: Instrument designs have certainly become more ergonomic with an increased focus on overall functionality and ease of use. Another key area of innovation has been through utilizing instruments intended for a specific patient with the ultimate goal of achieving implant fit optimization. These patient-matched instruments continue to evolve in our industry with continuing efforts in place to grow this market segment. Ergonomics will continue to be a leading effort when it comes to overall instrument design. From an instrument manufacturing perspective, we have seen an increased concentration in technology based systems, for example, direct metal laser sintering (DMLS). This fully automated system builds complex geometries layer by layer using a laser beam to melt fine metal powder. Tecomet has invested in this capability due to the multiple advantages inherent with this technology. Overall, DMLS allows Tecomet to provide our customers an economic manufacturing alternative leading to the earlier identification of design or manufacturing challenges.
Neenan: Less complicated instruments are headed in a more commercial or cost reduced, disposable direction. These instruments are now generally being packaged individually and are designed for a single use. UDI [unique device identification] implementation is another factor that our industry is working on implementing for implants, instruments, cases and trays. Low-cost implants is another trend we are seeing develop, low-cost suppliers are taking advantage of implant patent protection ending, submitting their own 510(k) for that product and offering it to the market for in some cases 40-50 percent less than market price. Market conditions that have made this possible are the transition of the larger surgical centers moving to centralized buying versus surgeons themselves being able to order what they prefer. The role of the OEM surgical rep has also been reduced in these cases due to the fact that the larger surgical centers have resident procedural experts because of their procedural volume.
Schultz: Reusable instruments are expensive and hard to clean and maintain. The impact of surgical site infection and hospital-acquired infections have been broadly reported. Reusable instruments, whether torque-limiters, drivers, awls, drill guides or depth gauges, pose a huge operations and maintenance burden on the P&L and balance sheet of every implant OEM. This is a $2 billion annual burden on the market. Over 90 percent of instruments that OEMs manufacture or purchase from suppliers are loaners or on consignment and 20 percent of those require spares or replacements every year. It’s a big problem that has created active pain across the OEM market.
Barbella: What is the biggest driver of orthopedic instrumentation innovation?
Buck: Set consolidation, compression of the number of instruments in a given set, materials, time on the table, instrument sets embracing more implant families. And cost, cost, cost. Take DePuy Synthes and the Attune system, for example. It’s a marquee product for Johnson & Johnson, probably one of the finest products ever fielded in the marketplace for a recon application. It’s brilliant in theory but the rate of adoption by the surgeons in the field has not been at the rate at which J&J expected. You have a beautiful system in Attune, but the surgeons aren’t necessarily adopting it at the rate at which they need to. I think it’s hard to differentiate today in Recon—hips and knees—the behavior to a large extent is a reflection of a commodity market for Recon. And certainly screws behave in the same manner, as do plates and rods in the spine market.
I don’t think there is enough demonstrated differentiation in procedural outcomes in the face of amplified cost pressures. So surgeons are sticking with what they know, at least this generation of surgeons are. I think contemplated procedural evolution through MAKO-style applications, for example, the robotic approach, are going to continue to attract and dramatically change procedural doctrine over time. But I think a lot of surgeons are sticking with what they know today versus the adoption or adaptation of a new generation of instrument sets. Over time, that will level out and the rate of adoption will find its own balance. But currently, that’s our reality. Economics have truly caught up with the orthopedic industry, we’re at a crossroads where ideology and reality have collided. There’s a lot of great ideology but can it actually manifest itself in reality? That’s where we’re at in the orthopedic industry today.
Differentiation is going to have to be pursued through instrument set configuration and the manner in which bundled services support both cases and tray sets or delivery systems and the client while managing their logistics for them. All of these marketplace activities are in the spirit of pursuing greater turns per instrument set, less sets in the field, with less time on the table leading to overall cost compression. Economics is forcing all of these behaviors in our marketplace.
Kemp: This can be defined in two areas – one being cost containment and the other being designing instruments that ultimately save overall time in the OR. These designs can either be made to be more efficient during use or considering ways to reduce the instrument set scope list by reducing the overall number of SKU’s. Overall cost containment pressures will continue to evolve in our industry based on increased demands from our customer base.
Neenan: MI surgical procedures.
Schultz: Current reusable instruments have high upfront costs; huge life cycle costs with spares, repairs and logistics; fall out of calibration; require ongoing reprocessing costs; pose infection risks; and require constant inventory management. Adoption of disposable instruments and kits have started with high procedure, low-risk implant procedures such as small bone and trauma implants. Rep-less operating rooms and disposable instruments and kits tailored for the outpatient market will dominate the market. Moving forward there will be a broad application of simplified instrument sets, fewer instruments per procedure and especially the adoption of single-procedure/disposable instrumentation.
Barbella: How are orthopedic instrument designs changing?
Buck: There’s a trend toward single-use instrumentation but that trend will be incremental. New materials are being explored along with the expanded use of over molding. OEMs are beginning to listen and embrace holistically DFM and feature-based pricing. I think that designs will be forced to achieve a pragmatic or practical dimensional reality. When you think about an instrument, when it goes through central sterile or a sterile processing department within a hospital or outpatient center and it has this high mirrored finish because someone in marketing within an OEM thought that’s what it should have, by the time it cycles one or two times through central sterile, that finish is gone. So why put it there to begin with? Let it have a more muted or matte finish, for example. Things like that will directly condition instrument design in the future, and should.
Also, a lot of instruments travel all around the country and they’re never used but yet they reside in a given set. OEMs and CMOs are being much smarter about set composition and the reduction of the total number of instruments and the total number of sets in the field. Sets, in terms of the manner in which they are configured, are becoming more efficient and innovation needs to be present in order to pursue that. Some instruments have by definition multiple uses in a given procedure. That needs to be more so. Some instruments have a very low usage in a given procedure. Those instruments need to be removed and/or that utility function needs to be added to another instrument. Weight is a consideration—weight means cost because of logistics, instrument density conditions, sterilization efficacy in both the sterile theatres and central processing areas of the hospitals. Set density and the number of sets associated with a given procedure have got to change. Innovation will drive that and economics will continue to force that. Everything is related to getting cost out of the value stream.
Kemp: Instrument designs are still considered to be somewhat specific to the project. However, from a change perspective, we are seeing instruments becoming longer in order to get deeper into the wound site. We’re definitely seeing a change—at least customers have asked, ‘is there a way to make that retractor, for example, longer or deeper?’
Neenan: More complex, more components, more challenging geometries and closer tolerances.
Schultz: Simplicity of design and robust operation. Instruments must first provide clinical value but must also provide economic value. We are seeing 10- and 20-year-old designs that have, for example, driver tips that require exacting geometry and tolerances and hence prove very difficult and expensive to machine. In some cases, they are over-engineered. They are designed that way to permit reusable instruments to mate with implant screws and connectors properly after many cycles of operation. The application of single-procedure instruments eliminates that cost and risk. They allow potentially relaxed tolerances while meeting clinical needs but at much lower price points while eliminating all reprocessing costs. It’s the way of the future for high-volume, low complexity implant procedures.
Horwood’s umbrage is clearly evident as he informs patients of his decision, blaming the move on a Cleveland Clinic directive to use joint replacement parts made only by Stryker Corp. or Zimmer Holdings Inc. In his letter late last summer, Horwood—an orthopedic surgeon—avoids critiquing the two companies’ implant quality, but hints that switching allegiances from DePuy products could negatively impact treatment.
“The Cleveland Clinic Foundation has instituted cost-cutting measures which limit the implant options for joint replacement surgery to just two vendors...Stryker and Zimmer,” Horwood’s letter read. “Dr. Horwood has 28 years of experience with DePuy J&J implants which, in his hands, have resulted in excellent clinical outcomes. Dr. Horwood does not feel in good conscience that he can jeopardize his patients by changing to an unfamiliar implant system simply to save money for the hospital system.”
Indeed, money played a key role in Horwood's breakup with the Cleveland Clinic. Physicians and hospital administrators have developed an adversarial relationship over the last half-decade as they’ve grappled with ways to provide quality healthcare at a lower cost. The challenge has frustrated both sides, though doctors have become particularly resentful of managers’ bottom-line tunnel vision.
Finances, however, may only be partly responsible for Horwood’s decision. Other likely contributors were product familiarity and, ultimately, resistance to change.
Physicians, like most human beings, are creatures of habit, preferring the familiar over the unknown. Most clinicians, for example, resisted the idea of electronic medical records in the late 1990s, deeming the time-saving tool an impersonal addition to their practices and an affront to the profession.
Many doctors have developed a similar mindset regarding medical products and equipment. Horwood, as he admitted in his letter, is a DePuy devotee; others are disciples of certain procedures (robotic surgery, for instance) or materials (trabecular metal, polyethylene, stainless steel, ceramics, etc.), and some have favorite tools.
Orthopedic surgeons have traditionally endorsed reusable instruments and surgical trays, but competition, healthcare reform, accuracy concerns, and—perhaps most importantly—cost, are engendering change in the operating room. Newly minted single-use, torque-limiting instruments and kits are becoming more prevalent as hospitals seek procedural efficiency, streamlined inventory, environmental harmony, and reduced cost.
While single-use instruments and kits have been a staple of many medical procedures for decades (cardiac rhythm management and neuromodulation, among others) they are a relatively new development in the orthopedic industry, particularly in the spinal sector.
Companies like Safe Orthopaedics, ECA Medical Instruments Inc. and Flower Orthopedics have developed single-use instruments for spinal surgery that promise to “eliminate unnecessary delays” as well as the need for expensive sterilization cycles. ECA’s disposable ratchet (the Model 300 Secure-Fast system) is designed to reduce surgical costs by more than $1,000 per procedure—a factor that will surely appeal to clinicians and payers striving to balance quality care and price value.
The global market for disposable medical supplies (instruments included) is forecast to grow 4.1 percent annually over the next two years, fueled by healthcare reform, an aging population, and a sharp focus on infection prevention. But convenience, cost, inventory management, and improved productivity will drive growth too, as more hospitals and surgeons realize their economic and clinical value.
To better understand the market forces responsible for single-use instrumentation as well as design trends and promising technological developments in surgical tools, Orthopedic Design & Technology spoke to a handful of manufacturing professionals and suppliers over the last few weeks. They included:
- Toby Buck, chairman/president/CEO/founder of Paragon Medical Inc., a Pierceton, Ind.-based Tier 1 supplier of custom and standard surgical instrument cases, trays and instruments, implantable components, and design and development services to the medical device industry. The company was incorporated in 1991.
- Mark Kemp, president/chief operating officer of Tecomet, a manufacturer of orthopedic implants, precision surgical instruments, trauma plates, and photochemically etched products for medical device customers. The company is headquartered in Wilmington, Mass.
- Jack Neenan, vice president of Sales & Business Development at Phillips Precision Medicraft Inc., an Elmwood Park, N.J.-based manufacturer of advanced orthopedic implants, instruments, sterilization delivery systems, cases, and trays.
- James B. Schultz, executive vice president of ECA Medical Instruments, a designer and manufacturer of single-use torque-limiting surgical instruments, fixed drivers and customized implant fixation kits to the medical device industry. The company is located in Thousand Oaks, Calif.
Michael Barbella: Please discuss the trends in orthopedic instrumentation and delivery systems.
Toby Buck: Trends that all contract manufacturing organizations (CMOs)—those like Greatbatch, Tecomet, Orchid—would say that the industry right now is under extreme stress because of downward cost pressures and that is directly affecting not only instruments but delivery systems, bundled services and implants. And that’s playing out obviously at our clients but it’s certainly trickled down and conditions the behavior of all of our income statements presently. So I think cost is the order of the day today, I think procurement organizations are the ones that are calling the shots internal to the OEMs out of necessity. I think all of us have to adjust and are adjusting to this context in a new operating paradigm. We have to not only continue to stay with the OEMs and serve them how they need to be served, but we have to get ahead of them to amplify our value proposition.
Mark Kemp: Tecomet is one of the largest contract manufacturers in the world and services several different market segments within the orthopedic industry. We continue to see the need for different levels of procedural innovation across all of these segments. To further address these opportunities, we offer multiple processing technologies that can result in value-added cost savings to our global customer base. Recently, our customers have increasingly requested the need for further instrument set cost reductions. Our Total Solution business approach attempts to manage these requests by working with the customer to reconfigure these sets to contain the essential core instruments. Additional innovation and cost reductions are being achieved via supply chain optimization opportunities and vendor-managed inventory is being further utilized.
Jack Neenan: MI (minimally invasive) surgical procedures are driving orthopedic instrument requirements. The benefit to MI procedures is less invasive and quicker recovery times for patients. On the case and tray side we are seeing some of our OEM customers looking to move away from using blue plastic wrap on vented cases and taking those same vented aluminum or stainless steel cases and putting them inside a “hard” pressure vented case. The “hard vented” case does away with the need for blue plastic wrap.
James Schultz: Each OEM implant firm wants to win more share of the fast-growth ambulatory surgery center (ASC) or outpatient market. It is very big and growing at 15 percent or more per year. Over 5,700 ASCs exist in the United States alone and 18 percent of their procedures are for orthopedics/spine. Who wants to go to a hospital? A day trip to the ASC is preferred and less expensive. More and more procedures are being approved yearly by the FDA [U.S. Food and Drug Administration] and payers are reimbursing these procedures at an increasing clip. OEMs will win if they have an implant solution that provides excellent outcomes and measured economic value.
The biggest issue facing every instrument manufacturer, implant OEM and end-user customer is cost. Each one is addressing how to drive out both upfront and life cycle costs to serve both the ASC and hospital market segments. Older business models are not sustainable. Moving forward there will be a broad application of lower-cost instruments and procedural kits, and especially the adoption of single-procedure/disposable instrumentation. Disposable instruments and procedure kits solve many active pain points today: They lower upfront and life cycle cost, reduce the risk of infection, are pristine and ready for surgery, curb inventory hassles, and are in perfect calibration to secure every orthopedic/spine implant.
Barbella: How has orthopedic instrumentation evolved over the last five to 10 years, and what major changes have you witnessed?
Buck: The orthopedic industry has learned from past sins of poor inventory management in the field and how that is supported not only logistically but also in the definition of launch quantities. I think the launches we experienced in the past as an industry with large hip and knee systems or large spine systems, for example, with instrumentation sets being deployed in the field, are not going to happen in the same manner they once did. The change is ever-present in terms of the cadence on launches for programs in the dominant segments we serve in orthopedics—hips, knees, spine, sports medicine, trauma and extremities. Over the last five to 10 years, there have been some major launches by all major OEMs but you have a parallel market implication associated with consolidation both at the OEM level and at the Tier 1 CMO level that has forced portfolio rationalization at the OEM level and, correspondingly, forced CMO consolidation to amplify the value proposition on the sourcing side of the fence for instrumentation, delivery systems and implants.
Another thing we need to do as an industry to get cost out of the game is…if you’re truly going to be a strategic partner, you have to agree between OEM and CMO, particularly with instrument applications, to embrace true design for manufacturing (DFM), true upstream feature-based pricing, and address the issues associated or attached to tolerancing that isn’t necessary in the actual utility application of the instrument or instrument set. You can get a tremendous amount of cost out through a pure application of DFM doctrine, feature-based pricing, and common sense applied to topographical tolerancing in instrumentation and delivery systems.
When you think about the configuration, the various topography, or the various dimensional characteristics of an instrument, there are radiuses and different features regarding the manner in which an instrument has tolerances that are associated with the finished machined product that are just too tight. The tighter a tolerance is—and this is common sense—it amplifies cost. There’s a parallel sensitivity that we all need to be aware of or mindful of as well—instrumentation today is treated in terms of regulatory compliance in the same manner that implants are. How can we expect as an industry to continue to impose a severe downward cost pressure on instrumentation and how can that coexist with amplified regulatory compliance, while CMS [the Center for Medicare and Medicaid Services] is compressing reimbursement thresholds? I don’t know how those things co-exist. And that’s causing the extreme downward cost pressure and stress on the industry that we’re experiencing right now. A related issue that has been coming for some time is less than effective instrument set inventory management in the field leading to too many sets in the field. Instrumentation is something that is not necessarily sold. Instrumentation is provided to the distribution infrastructure for the OEMs. Their distributors may or may not acquire some of it, but generally speaking instrumentation is treated as though it is tooling in the field for the OEM. That’s another part of the economic solution here that we all have to be sensitive to and ever-mindful of to help continue to find a way to amplify our value proposition to the marketplace as a CMO for integrated instrument set and bundled service sourcing.
Kemp: Instrument designs have certainly become more ergonomic with an increased focus on overall functionality and ease of use. Another key area of innovation has been through utilizing instruments intended for a specific patient with the ultimate goal of achieving implant fit optimization. These patient-matched instruments continue to evolve in our industry with continuing efforts in place to grow this market segment. Ergonomics will continue to be a leading effort when it comes to overall instrument design. From an instrument manufacturing perspective, we have seen an increased concentration in technology based systems, for example, direct metal laser sintering (DMLS). This fully automated system builds complex geometries layer by layer using a laser beam to melt fine metal powder. Tecomet has invested in this capability due to the multiple advantages inherent with this technology. Overall, DMLS allows Tecomet to provide our customers an economic manufacturing alternative leading to the earlier identification of design or manufacturing challenges.
Neenan: Less complicated instruments are headed in a more commercial or cost reduced, disposable direction. These instruments are now generally being packaged individually and are designed for a single use. UDI [unique device identification] implementation is another factor that our industry is working on implementing for implants, instruments, cases and trays. Low-cost implants is another trend we are seeing develop, low-cost suppliers are taking advantage of implant patent protection ending, submitting their own 510(k) for that product and offering it to the market for in some cases 40-50 percent less than market price. Market conditions that have made this possible are the transition of the larger surgical centers moving to centralized buying versus surgeons themselves being able to order what they prefer. The role of the OEM surgical rep has also been reduced in these cases due to the fact that the larger surgical centers have resident procedural experts because of their procedural volume.
Schultz: Reusable instruments are expensive and hard to clean and maintain. The impact of surgical site infection and hospital-acquired infections have been broadly reported. Reusable instruments, whether torque-limiters, drivers, awls, drill guides or depth gauges, pose a huge operations and maintenance burden on the P&L and balance sheet of every implant OEM. This is a $2 billion annual burden on the market. Over 90 percent of instruments that OEMs manufacture or purchase from suppliers are loaners or on consignment and 20 percent of those require spares or replacements every year. It’s a big problem that has created active pain across the OEM market.
Barbella: What is the biggest driver of orthopedic instrumentation innovation?
Buck: Set consolidation, compression of the number of instruments in a given set, materials, time on the table, instrument sets embracing more implant families. And cost, cost, cost. Take DePuy Synthes and the Attune system, for example. It’s a marquee product for Johnson & Johnson, probably one of the finest products ever fielded in the marketplace for a recon application. It’s brilliant in theory but the rate of adoption by the surgeons in the field has not been at the rate at which J&J expected. You have a beautiful system in Attune, but the surgeons aren’t necessarily adopting it at the rate at which they need to. I think it’s hard to differentiate today in Recon—hips and knees—the behavior to a large extent is a reflection of a commodity market for Recon. And certainly screws behave in the same manner, as do plates and rods in the spine market.
I don’t think there is enough demonstrated differentiation in procedural outcomes in the face of amplified cost pressures. So surgeons are sticking with what they know, at least this generation of surgeons are. I think contemplated procedural evolution through MAKO-style applications, for example, the robotic approach, are going to continue to attract and dramatically change procedural doctrine over time. But I think a lot of surgeons are sticking with what they know today versus the adoption or adaptation of a new generation of instrument sets. Over time, that will level out and the rate of adoption will find its own balance. But currently, that’s our reality. Economics have truly caught up with the orthopedic industry, we’re at a crossroads where ideology and reality have collided. There’s a lot of great ideology but can it actually manifest itself in reality? That’s where we’re at in the orthopedic industry today.
Differentiation is going to have to be pursued through instrument set configuration and the manner in which bundled services support both cases and tray sets or delivery systems and the client while managing their logistics for them. All of these marketplace activities are in the spirit of pursuing greater turns per instrument set, less sets in the field, with less time on the table leading to overall cost compression. Economics is forcing all of these behaviors in our marketplace.
Kemp: This can be defined in two areas – one being cost containment and the other being designing instruments that ultimately save overall time in the OR. These designs can either be made to be more efficient during use or considering ways to reduce the instrument set scope list by reducing the overall number of SKU’s. Overall cost containment pressures will continue to evolve in our industry based on increased demands from our customer base.
Neenan: MI surgical procedures.
Schultz: Current reusable instruments have high upfront costs; huge life cycle costs with spares, repairs and logistics; fall out of calibration; require ongoing reprocessing costs; pose infection risks; and require constant inventory management. Adoption of disposable instruments and kits have started with high procedure, low-risk implant procedures such as small bone and trauma implants. Rep-less operating rooms and disposable instruments and kits tailored for the outpatient market will dominate the market. Moving forward there will be a broad application of simplified instrument sets, fewer instruments per procedure and especially the adoption of single-procedure/disposable instrumentation.
Barbella: How are orthopedic instrument designs changing?
Buck: There’s a trend toward single-use instrumentation but that trend will be incremental. New materials are being explored along with the expanded use of over molding. OEMs are beginning to listen and embrace holistically DFM and feature-based pricing. I think that designs will be forced to achieve a pragmatic or practical dimensional reality. When you think about an instrument, when it goes through central sterile or a sterile processing department within a hospital or outpatient center and it has this high mirrored finish because someone in marketing within an OEM thought that’s what it should have, by the time it cycles one or two times through central sterile, that finish is gone. So why put it there to begin with? Let it have a more muted or matte finish, for example. Things like that will directly condition instrument design in the future, and should.
Also, a lot of instruments travel all around the country and they’re never used but yet they reside in a given set. OEMs and CMOs are being much smarter about set composition and the reduction of the total number of instruments and the total number of sets in the field. Sets, in terms of the manner in which they are configured, are becoming more efficient and innovation needs to be present in order to pursue that. Some instruments have by definition multiple uses in a given procedure. That needs to be more so. Some instruments have a very low usage in a given procedure. Those instruments need to be removed and/or that utility function needs to be added to another instrument. Weight is a consideration—weight means cost because of logistics, instrument density conditions, sterilization efficacy in both the sterile theatres and central processing areas of the hospitals. Set density and the number of sets associated with a given procedure have got to change. Innovation will drive that and economics will continue to force that. Everything is related to getting cost out of the value stream.
Kemp: Instrument designs are still considered to be somewhat specific to the project. However, from a change perspective, we are seeing instruments becoming longer in order to get deeper into the wound site. We’re definitely seeing a change—at least customers have asked, ‘is there a way to make that retractor, for example, longer or deeper?’
Neenan: More complex, more components, more challenging geometries and closer tolerances.
Schultz: Simplicity of design and robust operation. Instruments must first provide clinical value but must also provide economic value. We are seeing 10- and 20-year-old designs that have, for example, driver tips that require exacting geometry and tolerances and hence prove very difficult and expensive to machine. In some cases, they are over-engineered. They are designed that way to permit reusable instruments to mate with implant screws and connectors properly after many cycles of operation. The application of single-procedure instruments eliminates that cost and risk. They allow potentially relaxed tolerances while meeting clinical needs but at much lower price points while eliminating all reprocessing costs. It’s the way of the future for high-volume, low complexity implant procedures.