Michael Barbella, Managing Editor12.14.23
RevBio Inc. has been awarded a three-year grant from the National Institute on Aging (NIA), part of the National Institutes of Health, to further develop its TETRANITE technology.
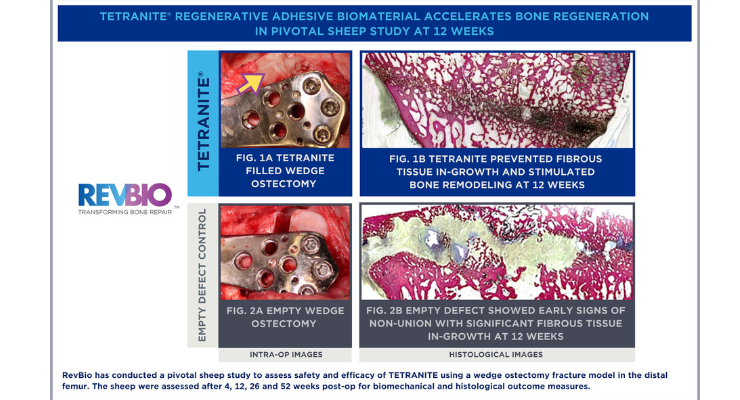
Research funded by this grant (up to $3.4 million) will focus on TETRANITE's ability to fill bone gaps, fixate bone fragments, and accelerate extremities fracture healing through its osteoconductive capabilities. TETRANITE will be used both as an interoperative fixation aid and as an adjunct to traditional hardware fixation to provide immediate load sharing between bone and metal plate and screw systems. By providing additional stability, TETRANITE will help achieve better healing and prevent complications and revision surgeries, RevBio executives claim. It can also be injected percutaneously as a minimally invasive stand-alone method of fixation to surgically treat fractures without the need for conventional open reduction internal fixation (ORIF). This grant will expand upon the pre-clinical research funded by a previous $2 million SBIR Phase II grant from the NIA.
“Our pre-clinical studies have demonstrated that TETRANITE is able to redistribute most of the load from plates and screws thereby reducing the likelihood of hardware failure and delayed healing,” said RevBio R&D Program Manager Brittany McDonough, who has led TETRANITE's product development. “This adhesive material also provides a resorbable scaffold for the regeneration of bone which accelerates healing within the first few weeks following a traumatic injury.”
Upper and lower extremity injuries equate to more than 40% of the approximate 2.1 million fragility fractures that occur annually in the United States.1,2 This number is expected to triple by 2040 as the population ages.3,4 External fixation, percutaneous pinning with K-wires, and conventional ORIF procedures that involve the use of plates and screws are interventions that are currently being used to treat these extremity fractures5 but despite advances in surgical technique and implant design, nonunion, malunion, and hardware failure continue to remain a significant cause of revision surgeries in the elderly.6 Complication rates as high as 36% have been reported and involve the onset of carpal tunnel syndrome, complex regional pain syndrome, tendon irritation and rupture, and deep infection.7,8 These complications can lead to difficulty performing basic daily tasks, loss of independence, reduction in quality of life, and increased mortality.9,10
“Fractures are often comminuted, and it is difficult to get the pieces back together and so having an adhesive would really help achieve an anatomic reduction,” said Michael J. Weaver, M.D., chief of Orthopaedic Trauma at Brigham & Women’s Hospital, and member of RevBio’s Orthopaedic Scientific Advisory Board. “This material is mechanically strong to allow early weight bearing which is something we cannot do currently in peri-articular fractures treated with metal implants. Having a material that can both augment fixation to prevent hardware failure and facilitate healing will fundamentally improve how our patients do.”
RevBio is a clinical stage medical device company in Lowell, Mass., that is developing and commercializing a patented, synthetic, injectable, self-setting, and osteoconductive bone adhesive biomaterial called TETRANITE. This material is designed for use in the dental, cranial, and broader orthopaedic markets as well as the animal health market. RevBio's Tetranite technology is not yet approved for commercial use.
References
1 https://www.aaos.org/aaosnow/2022/thursday/research/research12/
2 https://www.osteoporosis.foundation/facts-statistics/epidemiology-of-osteoporosis-and-fragility-fractures
3 Watts, Nelson B., et al. "National Osteoporosis Foundation 2008 Clinician's Guide to Prevention and Treatment of Osteoporosis and the World Health Organization Fracture Risk Assessment Tool (FRAX): what they mean to the bone densitometrist and bone technologist." Journal of clinical densitometry: the official journal of the International Society for Clinical Densitometry 11.4 (2008): 473-477.
4 Burge, Russel, et al. "Incidence and economic burden of osteoporosis‐related fractures in the United States, 2005–2025." Journal of bone and mineral research 22.3 (2007): 465-475.
5 Grewal R, MacDermid JC, King GJ, Faber KJ. Open reduction internal fixation versus percutaneous pinning with external fixation of distal radius fractures: a prospective, randomized clinical trial. J Hand Surg Am. 2011 Dec;36(12):1899-906. doi: 10.1016/j.jhsa.2011.09.015. Epub 2011 Nov 3. PMID: 22051229.
6 Kloen, Peter, Geert A. Buijze, and David Ring. "Management of forearm nonunions: current concepts." Strategies in trauma and limb reconstruction 7 (2012): 1-11.
7 Thorninger, Rikke, Mette Lund Madsen, Daniel Wæver, Lars Carl Borris, and Jan Hendrik Duedal Rölfing. "Complications of volar locking plating of distal radius fractures in 576 patients with 3.2 years follow-up." Injury 48, no. 6 (2017): 1104-1109.
8 Watts, Nelson B., et al. "National Osteoporosis Foundation 2008 Clinician's Guide to Prevention and Treatment of Osteoporosis and the World Health Organization Fracture Risk Assessment Tool (FRAX): what they mean to the bone densitometrist and bone technologist." Journal of clinical densitometry: the official journal of the International Society for Clinical Densitometry 11.4 (2008): 473-477.
9 Marchewka, Jakub, Jacek Głodzik, Wojciech Marchewka, and Edward Golec. "Higher Mortality in Men Compared with Women following Distal Radius Fracture in Population Aged 50 Years or Above: Are Common Distal Radius Fracture Classifications Useful in Predicting Mortality?" BioMed research international 2019 (2019).
10 Nellans, Kate W., Evan Kowalski, and Kevin C. Chung. "The epidemiology of distal radius fractures." Hand clinics 28, no. 2 (2012): 113-125.
Research funded by this grant (up to $3.4 million) will focus on TETRANITE's ability to fill bone gaps, fixate bone fragments, and accelerate extremities fracture healing through its osteoconductive capabilities. TETRANITE will be used both as an interoperative fixation aid and as an adjunct to traditional hardware fixation to provide immediate load sharing between bone and metal plate and screw systems. By providing additional stability, TETRANITE will help achieve better healing and prevent complications and revision surgeries, RevBio executives claim. It can also be injected percutaneously as a minimally invasive stand-alone method of fixation to surgically treat fractures without the need for conventional open reduction internal fixation (ORIF). This grant will expand upon the pre-clinical research funded by a previous $2 million SBIR Phase II grant from the NIA.
“Our pre-clinical studies have demonstrated that TETRANITE is able to redistribute most of the load from plates and screws thereby reducing the likelihood of hardware failure and delayed healing,” said RevBio R&D Program Manager Brittany McDonough, who has led TETRANITE's product development. “This adhesive material also provides a resorbable scaffold for the regeneration of bone which accelerates healing within the first few weeks following a traumatic injury.”
Upper and lower extremity injuries equate to more than 40% of the approximate 2.1 million fragility fractures that occur annually in the United States.1,2 This number is expected to triple by 2040 as the population ages.3,4 External fixation, percutaneous pinning with K-wires, and conventional ORIF procedures that involve the use of plates and screws are interventions that are currently being used to treat these extremity fractures5 but despite advances in surgical technique and implant design, nonunion, malunion, and hardware failure continue to remain a significant cause of revision surgeries in the elderly.6 Complication rates as high as 36% have been reported and involve the onset of carpal tunnel syndrome, complex regional pain syndrome, tendon irritation and rupture, and deep infection.7,8 These complications can lead to difficulty performing basic daily tasks, loss of independence, reduction in quality of life, and increased mortality.9,10
“Fractures are often comminuted, and it is difficult to get the pieces back together and so having an adhesive would really help achieve an anatomic reduction,” said Michael J. Weaver, M.D., chief of Orthopaedic Trauma at Brigham & Women’s Hospital, and member of RevBio’s Orthopaedic Scientific Advisory Board. “This material is mechanically strong to allow early weight bearing which is something we cannot do currently in peri-articular fractures treated with metal implants. Having a material that can both augment fixation to prevent hardware failure and facilitate healing will fundamentally improve how our patients do.”
RevBio is a clinical stage medical device company in Lowell, Mass., that is developing and commercializing a patented, synthetic, injectable, self-setting, and osteoconductive bone adhesive biomaterial called TETRANITE. This material is designed for use in the dental, cranial, and broader orthopaedic markets as well as the animal health market. RevBio's Tetranite technology is not yet approved for commercial use.
References
1 https://www.aaos.org/aaosnow/2022/thursday/research/research12/
2 https://www.osteoporosis.foundation/facts-statistics/epidemiology-of-osteoporosis-and-fragility-fractures
3 Watts, Nelson B., et al. "National Osteoporosis Foundation 2008 Clinician's Guide to Prevention and Treatment of Osteoporosis and the World Health Organization Fracture Risk Assessment Tool (FRAX): what they mean to the bone densitometrist and bone technologist." Journal of clinical densitometry: the official journal of the International Society for Clinical Densitometry 11.4 (2008): 473-477.
4 Burge, Russel, et al. "Incidence and economic burden of osteoporosis‐related fractures in the United States, 2005–2025." Journal of bone and mineral research 22.3 (2007): 465-475.
5 Grewal R, MacDermid JC, King GJ, Faber KJ. Open reduction internal fixation versus percutaneous pinning with external fixation of distal radius fractures: a prospective, randomized clinical trial. J Hand Surg Am. 2011 Dec;36(12):1899-906. doi: 10.1016/j.jhsa.2011.09.015. Epub 2011 Nov 3. PMID: 22051229.
6 Kloen, Peter, Geert A. Buijze, and David Ring. "Management of forearm nonunions: current concepts." Strategies in trauma and limb reconstruction 7 (2012): 1-11.
7 Thorninger, Rikke, Mette Lund Madsen, Daniel Wæver, Lars Carl Borris, and Jan Hendrik Duedal Rölfing. "Complications of volar locking plating of distal radius fractures in 576 patients with 3.2 years follow-up." Injury 48, no. 6 (2017): 1104-1109.
8 Watts, Nelson B., et al. "National Osteoporosis Foundation 2008 Clinician's Guide to Prevention and Treatment of Osteoporosis and the World Health Organization Fracture Risk Assessment Tool (FRAX): what they mean to the bone densitometrist and bone technologist." Journal of clinical densitometry: the official journal of the International Society for Clinical Densitometry 11.4 (2008): 473-477.
9 Marchewka, Jakub, Jacek Głodzik, Wojciech Marchewka, and Edward Golec. "Higher Mortality in Men Compared with Women following Distal Radius Fracture in Population Aged 50 Years or Above: Are Common Distal Radius Fracture Classifications Useful in Predicting Mortality?" BioMed research international 2019 (2019).
10 Nellans, Kate W., Evan Kowalski, and Kevin C. Chung. "The epidemiology of distal radius fractures." Hand clinics 28, no. 2 (2012): 113-125.